
Molecular sieves, used to remove carbon dioxide, are materials with extremely small pores. These pores trap and separate carbon dioxide molecules from gas streams. Using molecular sieves helps ensure pipeline safety and maintain natural gas quality.
| Aspect | Explanation |
|---|---|
| Pipeline Integrity | CO2 can mix with water and make carbonic acid. This acid can damage pipelines and make them unsafe. |
| Natural Gas Specifications | CO2 is a contaminant that lowers the BTU value of natural gas. Removing it is important. |
Taking out CO2 also helps slow down climate change. It helps industries use cleaner technology.
Key Takeaways
- The small pore size of molecular sieves allows them to trap carbon dioxide, thus ensuring pipeline safety and improving natural gas quality.
- There are various types of molecular sieves, such as 13X and 5A. Each type of molecular sieve has its specific pore size. The size of the pore determines its efficiency in capturing carbon dioxide.
- Using molecular sieves contributes to environmental protection. They can reduce greenhouse gas emissions and purify the air.
- Molecular sieves require regular maintenance to ensure proper functioning and extend their carbon dioxide capture lifespan.
- Industry uses molecular sieves to achieve cleaner production processes, helping them comply with stringent environmental regulations.
What Are Molecular Sieves?

Definition and Structure
A molecular sieve is a special material that can trap certain specific molecules. They have tiny pores of the same size. These pores allow some molecules to pass through but trap others. Scientists use materials such as aluminosilicates or carbon to create molecular sieves. If viewed up close, they look like a honeycomb. Each pore acts like a miniature trap, capturing the appropriate molecules.
The size of the pores determines which gases are trapped. The table below shows the effect of different pore sizes on carbon dioxide capture:
| Molecular Sieve | Pore Size (Å) | Adsorption Performance for CO2 |
|---|---|---|
| 13X | 8–10 | Better due to strong polarity |
| 5A | 5 | Lower adsorption capacity |
| 4A | 4 | Lower adsorption capacity |
| 3A | 4 | Lower adsorption capacity |
If there are more holes, the sieve can catch more carbon dioxide. This is why molecular sieves are important for cleaning air and capturing carbon.
How Molecular Sieves Work for CO2 Removal
Molecular sieves utilize their unique pore size and chemical properties to capture carbon dioxide. When a gas mixture passes through a molecular sieve, only molecules matching the pore size are captured. Carbon dioxide is easily captured by some molecular sieves, especially those with larger pore sizes like 13X.
Carbon dioxide has a strong polar quadrupole moment. This helps it adsorb onto the inner wall of the molecular sieve. Molecular sieves have a higher adsorption capacity for carbon dioxide than for other gases. For example, 13X and 5A type molecular sieves are particularly adept at capturing carbon dioxide, which helps reduce greenhouse gas emissions.
Industry uses these molecular sieves to remove carbon dioxide, for example, to purify natural gas, air, and factory emissions. This helps protect the environment and reduce pollution. Molecular sieves are crucial for air purification and carbon capture.
Note: Molecular sieves are a dependable way to catch and separate gases. They are important for helping the planet.
Types of Molecular Sieves for Carbon Dioxide Removal
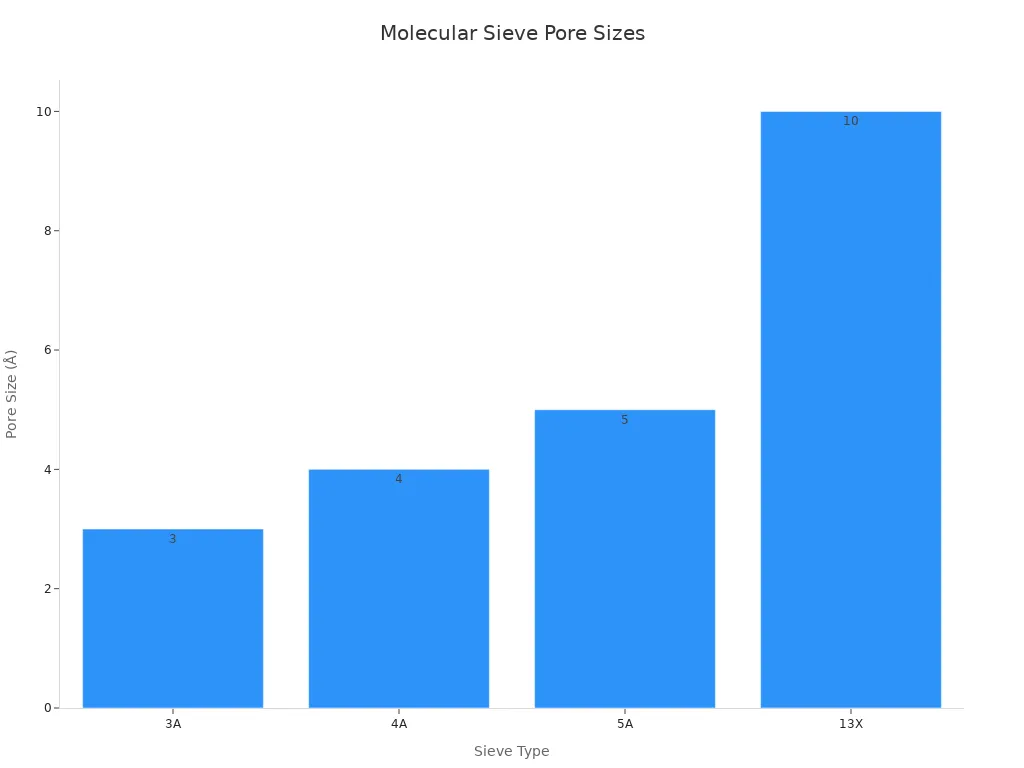
Molecular sieves help remove carbon dioxide from gases. Each sieve has its own shape and hole size. This changes how well it can catch carbon dioxide. The table below shows the main types, their hole sizes, and uses:
| Type | Pore Size (Å) | Main Applications |
|---|---|---|
| 3A | 3 | Adsorbs water and polar molecules; used in petroleum. |
| 4A | 4 | Adsorbs water and CO2; used in natural gas industry. |
| 5A | 5 | Adsorbs larger molecules; used in air separation. |
| 13X | 10 | Adsorbs larger molecules; used in gas purification. |
Zeolite 13X and 5A
Zeolite 13X and 5A are commonly used sieves for carbon dioxide removal. Zeolite 13X has a relatively large pore size of approximately 10 angstroms, allowing it to adsorb more carbon dioxide than other sieves. At 20°C, zeolite 13X can adsorb 1.79 mmol of carbon dioxide per gram. At lower temperatures, such as -80°C, its adsorption capacity can reach 5.38 mmol per gram. Sometimes, the adsorption capacity of zeolite 13X can even reach around 3.8 mmol per gram.
Zeolite 13X has a better adsorption capacity for carbon dioxide than other gases, especially nitrogen and methane. The isothermal heat of adsorption for carbon dioxide is 37.2 kJ/mol, for nitrogen it is 12.8 kJ/mol, and for methane it is 15.3 kJ/mol. This means that zeolite 13X is very suitable for gas purification and carbon dioxide capture. Zeolite 5A has a pore size of 5 angstroms. It can also capture carbon dioxide, but it is most effective for larger molecules. It is often used for air separation.
4A Molecular Sieve
4A molecular sieves have a pore size of 4 angstroms. They are widely used in the natural gas industry and air purification systems. These molecular sieves can capture water and carbon dioxide. In air separation units, 4A molecular sieves can remove water and carbon dioxide from the air. This prevents clogging of cryogenic heat exchangers. Carbon dioxide can freeze and cause problems.
4A molecular sieves are ideal for purifying gases and air. They can keep natural gas dry and remove carbon dioxide. This protects equipment and improves gas quality. These molecular sieves help reduce greenhouse gas emissions and help industries comply with regulations.
Carbon Molecular Sieves Overview
Carbon molecular sieves (CMS) are advanced molecular sieves used for removing carbon dioxide. They excel when high selectivity and efficiency are required. Carbon molecular sieves (CMS) have a large surface area and uniform pore size, which contributes to their rapid and efficient capture of carbon dioxide. By modifying their internal structure and chemical composition, manufacturers can optimize the performance of carbon molecular sieves for specific applications.
| Structural Feature | Impact on CO2 Removal Efficiency |
|---|---|
| Large Surface Area | Maximizes interaction with CO2, enhancing capacity to capture CO2. |
| Uniform Pore Structure | Provides high efficiency in capturing CO2 from gas streams. |
YUANHAO’s carbon molecular sieves are a prime example of this technology. Designed specifically for separating gases such as carbon dioxide and nitrogen, they can quickly and efficiently separate pure carbon dioxide. YUANHAO’s carbon molecular sieves help various industries reliably remove carbon dioxide, effectively reducing carbon emissions and purifying the air.
Carbon molecular sieves are also used in air filters, helping industries reduce greenhouse gas emissions and achieve their goals. Using carbon molecular sieves helps companies improve carbon capture efficiency and achieve a cleaner working environment.
Tip: Choose the appropriate molecular sieve based on the gas type, required purity, and application scenario. Each molecular sieve has its unique advantages in removing carbon dioxide.
CO2 Removal Mechanisms
Adsorption Process
Molecular sieves capture carbon dioxide from airflow using adsorption. These materials act like miniature sponges, trapping and retaining carbon dioxide within their pores. This technology plays a vital role in many industries, such as natural gas purification and refining. It helps reduce greenhouse gas emissions and supports green environmental goals.
- Excellent at removing carbon dioxide from plants
- Aids in purifying and refining natural gas
- Beneficial for environmental protection and improves work efficiency
Molecular sieves have an extremely strong adsorption capacity for carbon dioxide. They can capture carbon dioxide even in the presence of other gases. This makes them crucial in carbon capture technologies. Many companies use molecular sieves in air filters to keep the air clean and safe.
Thermal Regeneration
After capturing carbon dioxide, molecular sieves need to release it to function again. This step is called thermal regeneration. The molecular sieve is heated to approximately 120°C. The high temperature breaks the binding force between the molecular sieve and the gas, thus releasing the carbon dioxide from the molecular sieve.
Thermal Variable Frequency Adsorption (TSA) utilizes hot air or gas to assist in this step. As the temperature rises, the screen’s ability to adsorb carbon dioxide decreases, the gas escapes, and the screen can be reused. The energy required depends on the type of screen and the water content in the gas.
Factors Affecting Performance
Many things can change how well molecular sieves catch carbon dioxide and how long they last. The table below shows some important things:
| Key Factors | Description |
|---|---|
| Stability | Molecular sieves have strong chemical properties, so they are good for carbon dioxide removal. |
| Selectivity | 13X and 5A molecular sieves are very good at picking carbon dioxide. |
| Capacity | These sieves can hold a lot of carbon dioxide. |
| Resistance | They can handle heat and wear, so they work well in many places. |
| Production | They are easy to make in big amounts, so factories can use them. |
Other things include pressure, flow speed, temperature, and humidity. If there are too many other things in the gas, the pores can get blocked and not catch as much gas. Using the sieves too much can also make them not last as long. Physical stress, like cracking or squeezing, and chemicals can hurt the sieves and make them not work as well. Companies need to check these things to keep their systems working and to lower carbon emissions.
Tip: Check and take care of molecular sieves often. This helps them catch more carbon dioxide and last longer.
Applications of Carbon Dioxide Removal Sieves

Natural Gas Treatment
Natural gas must be extremely clean before entering pipelines. Carbon dioxide removal sieves help accomplish this. The pores of 5A molecular sieves can trap carbon dioxide and water vapor. Larger molecules can pass through the pores without being trapped. This makes natural gas more suitable for heating and prevents pipelines from rusting. These molecular sieves are reusable. Workers clean them by heating or changing the pressure, selectively capturing specific gases. This makes natural gas safer and more practical.
Air Purification
Factory and building facilities require clean air to ensure personnel safety. Molecular sieves can capture carbon dioxide from the air. They have a large surface area and can capture certain specific gases. This makes them excellent in air purification systems. These molecular sieves are more effective, safer, and more economical than other materials.
- Molecular sieves are highly efficient at capturing carbon dioxide.
- They work better than activated carbon in factories.
- Their special properties make them suitable for a variety of gases.
Medical and Industrial Uses
Hospitals and factories require pure gases to operate properly. Carbon dioxide removal sieves assist air separation devices by capturing carbon dioxide and water vapor. They also help purify hydrogen and produce oxygen. These sieves can capture harmful gases, thus ensuring the safe and smooth operation of the machine.
Note: Using carbon dioxide removal sieves is important for safety, good work, and helping the planet.
Molecular Sieves Benefits and Limitations
Advantages of Molecular Sieves
For businesses that need to capture carbon dioxide, molecular sieves offer numerous advantages. They have a large surface area, which helps capture significant amounts of carbon dioxide from the gas. Therefore, plants can meet stringent air quality standards. The table below lists some of the main advantages:
| Advantage | Description |
|---|---|
| Large Surface Area | Lets CO2 touch the sieve more, so it catches more carbon dioxide. |
| Efficient Adsorption | Helps grab and separate CO2 before it gets into the air, which is good for the environment. |
| Selective Nature | 5A molecular sieves catch CO2 but let other gases go by, so removal works well. |
Molecular sieves also help with carbon capture and storage. They lower greenhouse gases, which is good for Earth. Many companies use them because they can catch water and dirt, making products very pure. This is very important when drying natural gas, where low dew points are needed.
Molecular sieves save money. They use special adsorption by size and polarity, so they catch carbon dioxide using less energy. They can be used again without chemicals, which is better for the planet.
Common Challenges
Molecular sieves work well, but they have some problems. Water and other things can make it harder for them to catch carbon dioxide. For example, 13X molecular sieves catch water vapor. This keeps air dry and protects machines.
Some common problems are:
- Equipment can rust when carbon dioxide and water make carbonic acid.
- Heat exchangers can get dirty from solid stuff, which makes them work less well and need cleaning.
- Product quality can go down if carbon dioxide hurts catalysts or makes separation harder.
Molecular sieves must catch both water and carbon dioxide to stop problems at cold temperatures, like solid CO2 forming. Checking and taking care of the sieves often helps everything work well and keeps carbon dioxide capture strong.
Tip: Workers should look at molecular sieves often to make sure they keep catching carbon dioxide well.
Molecular sieves are important in CO2 removal systems. They use their ability to grab carbon dioxide to catch gas in many places. The table below shows how well they work:
| Industry/Application | Molecular Sieve Type | CO2 Removal Effectiveness |
|---|---|---|
| Air Separation Units | 13X | Purities up to 93% ±3% |
| Medical Oxygen Production | 13X | High purity oxygen output |
Molecular sieves pick certain gases, save energy, and help the environment. They help with carbon capture and storage, so companies like to use them. New technology and strict rules make more companies use sieves. Molecular sieves will keep catching CO2 as industries look for better ways.
- Molecular sieves help lower pollution.
- They use less energy than chemical ways.
- Their strong features let them catch lots of CO2.
Companies should use molecular sieves for safe and good CO2 capture in modern removal systems.



